Driving the field of LPEM forward at the Gordon conference

We realized that most researchers were still assuming that all liquid holders for LPEM are still relying on the ‘bathtub’ style (i.e. pocket structure where the 2 chips are placed). This is far from ideal, as the liquid bypasses the nano-cell and it only flows towards the window by diffusion in a non-controlled and spontaneous way. Therefore, it was a big relief for the LPEM community to learn that our Stream system now enables the real benefits, like (a) accurately controlling pressure and flow over the window, (b) controlling membrane bulging (i.e. controlling the liquid thickness) to enable higher resolutions, (c) enabling meaningful results in structure determination and analytical microscopy studies (e.g. EDS, EELS, electron diffraction), (d) controlling and mitigating bubble formation and most importantly, (e) reproducible experiments.
The Gordon Research Conferences are a special type of conference aimed at advancing frontier research. The idea is to bring all the relevant people in the field together to discuss and present (unpublished) results and to talk about the future directions of the field. All the major players in the field were present, and there was a lot of time for interaction. This created an open atmosphere, in which knowledge was shared and collaborations were established.
It became clear that the Liquid Phase Electron Microscopy community is maturing. LPEM offers a unique way for scientists to obtain information within a wide range of fields, including nanoparticle synthesis, self-assembly, corrosion, batteries, semicon, proteins and cells. However, compared to Cryo-EM, the field is still in its early days. A number of challenges still exist before results will be reproducibly accepted by non-microscopist communities. For example how to deal with the influence of the e-beam and how to control other influencing parameters.
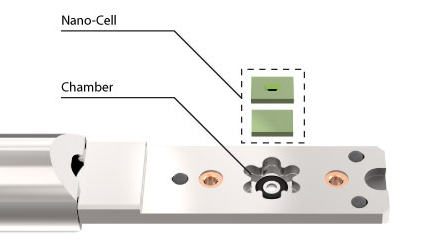
‘Bathtub’ style LPEM system. Liquid bypasses the Nano-Cell and flows toward the window in a non-controlled and spontaneous way.
DENSsolutions Stream LPEM system. On-chip microfluidic channel enables full control over the liquid flow and pressure, thus the liquid-sample interaction.
Thank you for reading
To learn more about our LPEM system: